Investigating the effect of substrate concentration on enzyme activity
Investigation into the effect of substrate concentration on enzyme activity is part of the Component 1 specification (Biological reactions are regulated by enzymes). You should read over the theory of enzyme structure as well as the effect of substrate concentration on enzyme activity from the drop down menu above.
Introduction
The substrate concentration in an enzyme catalysed reaction affects how much product is made. Catalase is an enzyme that increases the breakdown of hydrogen peroxide, a toxin produced as a bi-product of metabolic reactions in cells. The reaction of catalase is shown below.
2H2O2 -----> 2H2O + O2
Catalase is found in all tissue samples such as liver, apple and potato tissue. The liver sample would contain the highest concentration of catalase because these liver cells are more metabolically active than apple or potato (so the liver produces more hydrogen peroxide that it will then need to break down to prevent it becoming toxic). However, relatively high concentrations in raw potatoes are sufficient for use in this practical.
Apparatus and reagents
Freshly cut potato cylinders
Pestle and mortar
Specimen tubes/test tubes
Stock solution of hydrogen peroxide 100% volume/volume (labelled stock solution)
Filter paper discs
Forceps
Stopwatch
Syringe
Distilled water
Paper towel
Method
1. Grind a 2 cm piece of potato cylinder with 5 mL of distilled water to make a smooth paste containing catalase enzyme
2. Place 10 mL of H2O2 in a specimen tube/ test tube
3. Using forceps, dip a filter paper disc into the enzyme suspension, tap off the excess
4. Gently place the filter paper disc at the bottom of the hydrogen peroxide solution using the forceps and start the stopwatch at exactly the same time as you release the disc using the forceps
5. Measure the time, to the nearest 0.1 second, that it takes for the disc to float up to the surface of the H2O2 solution
6. Remove the disc from the tube using forceps and discard.
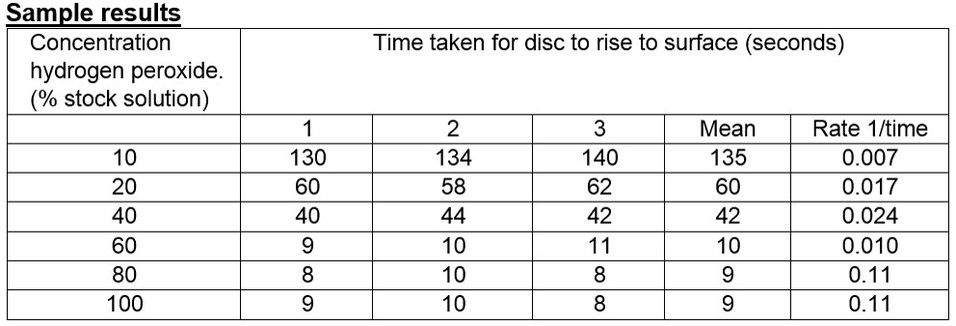
7. A serial dilution of the original 100% H2O2 stock solution should be prepared to generate a series of different substrate concentrations (recommended: 80%, 60%, 40%, 20% and 10%).
8. Repeat steps 1-6 with each of the new dilutions making sure there are three measurements taken (i.e. three discs are used) for each substrate concentration.;
Investigation into the effect of substrate concentration on enzyme activity is part of the Component 1 specification (Biological reactions are regulated by enzymes). You should read over the theory of enzyme structure as well as the effect of substrate concentration on enzyme activity from the drop down menu above.
Introduction
The substrate concentration in an enzyme catalysed reaction affects how much product is made. Catalase is an enzyme that increases the breakdown of hydrogen peroxide, a toxin produced as a bi-product of metabolic reactions in cells. The reaction of catalase is shown below.
2H2O2 -----> 2H2O + O2
Catalase is found in all tissue samples such as liver, apple and potato tissue. The liver sample would contain the highest concentration of catalase because these liver cells are more metabolically active than apple or potato (so the liver produces more hydrogen peroxide that it will then need to break down to prevent it becoming toxic). However, relatively high concentrations in raw potatoes are sufficient for use in this practical.
Apparatus and reagents
Freshly cut potato cylinders
Pestle and mortar
Specimen tubes/test tubes
Stock solution of hydrogen peroxide 100% volume/volume (labelled stock solution)
Filter paper discs
Forceps
Stopwatch
Syringe
Distilled water
Paper towel
Method
1. Grind a 2 cm piece of potato cylinder with 5 mL of distilled water to make a smooth paste containing catalase enzyme
2. Place 10 mL of H2O2 in a specimen tube/ test tube
3. Using forceps, dip a filter paper disc into the enzyme suspension, tap off the excess
4. Gently place the filter paper disc at the bottom of the hydrogen peroxide solution using the forceps and start the stopwatch at exactly the same time as you release the disc using the forceps
5. Measure the time, to the nearest 0.1 second, that it takes for the disc to float up to the surface of the H2O2 solution
6. Remove the disc from the tube using forceps and discard.
7. A serial dilution of the original 100% H2O2 stock solution should be prepared to generate a series of different substrate concentrations (recommended: 80%, 60%, 40%, 20% and 10%).
8. Repeat steps 1-6 with each of the new dilutions making sure there are three measurements taken (i.e. three discs are used) for each substrate concentration.;
Risk assessment
Hazard > Risk of hazard causing harm > Control measure to limit risk
For example:
Cork borers are sharp
Cork borers may cut skin when cutting cylinders
The cylinders of tissue must be cut on a white tile with the force directed downwards
Hydrogen peroxide is an irritant to skin and eyes
Hydrogen peroxide may splash onto the skin or into the eyes
Wear appropriate personal protective equipment (P.P.E) such gloves, lab coats and goggles
Further work
• Extension work could include a comparison of catalase activity from a range of sources such as mung bean or liver.
• The technique could be used to study the effect of substrate or enzyme concentration on enzyme activity.
• Students should be encouraged to calculate the rate of reaction (1/time).
Practical techniques
• Use appropriate apparatus to record a range of quantitative measurements (to include mass, time, volume, temperature, length and pH)
• Use laboratory glassware apparatus for a variety of experimental techniques to include serial dilution
Hazard > Risk of hazard causing harm > Control measure to limit risk
For example:
Cork borers are sharp
Cork borers may cut skin when cutting cylinders
The cylinders of tissue must be cut on a white tile with the force directed downwards
Hydrogen peroxide is an irritant to skin and eyes
Hydrogen peroxide may splash onto the skin or into the eyes
Wear appropriate personal protective equipment (P.P.E) such gloves, lab coats and goggles
Further work
• Extension work could include a comparison of catalase activity from a range of sources such as mung bean or liver.
• The technique could be used to study the effect of substrate or enzyme concentration on enzyme activity.
• Students should be encouraged to calculate the rate of reaction (1/time).
Practical techniques
• Use appropriate apparatus to record a range of quantitative measurements (to include mass, time, volume, temperature, length and pH)
• Use laboratory glassware apparatus for a variety of experimental techniques to include serial dilution